In a study published on January 24, 2022 in the journal Molecular Psychiatry, the scientific team “Central Control of Eating Behavior and Energy Expenditure” (Functional and Adaptive Biology Unit – Université Paris Cité/CNRS) discovered a new mechaninsm which, by linking the gut and the brain, control appetite. This mechanism could be at the origin of compulsive eating disorders, such as binge-eating. By focusing on this mechanism using molecules targeting the endocannabinoid system, the scientists are developing new approaches that may have a relevant therapeutic impact.
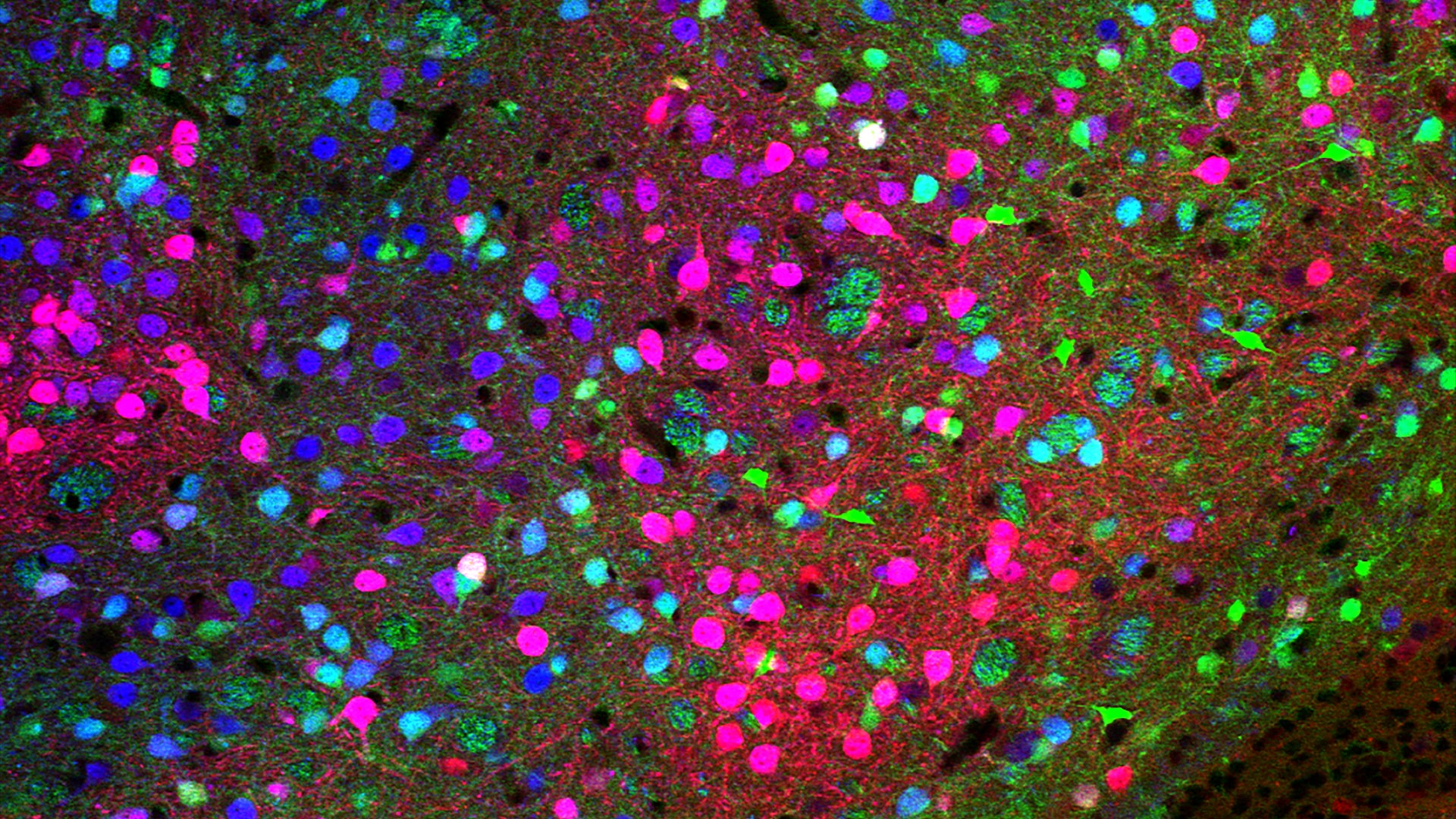
Noyau accumbens
Confocal microscopy image showing a region of the nucleus accumbens, a brain structure that is part of the dopamine circuit and highly involved in reward processes.
© Dr. Giuseppe Gangarossa
The brain regulates food intake in two main ways. The first takes into account energy demands that an organism requires for its survival. This is mostly governed by the hypothalamus, which dictates the feelings of hunger and satiety. However, food intake is also governed by cravings or the desire to eat. This craving involves another system of the brain mainly associated with the release of neurotrasmitter dopamine: the reward system. These two regulatory circuits coexist and permanently communicate, and toghether govern the basic structure of food intake: I eat to survive and I eat what I choose.
A New Regulator: The Gut-Brain Axis
For a long time, it has been well-accepted that these two circuits worked in parallel and managed the different components of food intake. Today, researchers are discovering a new mechanism regulating food intake, which is not located in the brain but in the periphery: the gut-brain axis, and more specifically one of its components, the vagus nerve. The vagus nerve is the longest nerve in the body. The neurons contained in its ganglia ensure the interoceptive relay, i.e. communication between the brain and the viscera (stomach, liver, intestine, pancreas) and viceversa.
In the study conducted by Dr. Chloe Berland and directed by Dr. Giuseppe Gangarossa, an associate professor at Université Paris Cité, the researchers of “Central Control of Eating Behavior and Energy Expenditure” team worked with mice whose behavior is similar to eating disorders associated with binge-eating. These mice, in addition to their usual food, were also given the opportunity to eat palatable food (rich in fat and sugar) every day, but for only one hour. By giving the mice access to unlimited regular food, the scientists were able to focus primarily on the brain circuits underlying the control of hedonic food intake.
The scientists progressively observed that the mice ate more and more of the palatable food and less and less of their regular food pellets. This phenotype led to the emergence of a compulsive eating disorder, binge-eating. Everyday, the mice waited impatiently for the palatable food. This resulted in a significant change in their physiological and metabolic states as well as in an increase in dopamine, the reward-associated transmitter; all of which reinforced the compulsive binge-eating behavior.
However, it would be too simplistic to consider this compulsive behavior as the result of only brain’s actions, since the mechanism bringing nutritional information to the brain is extensive and begins in the gastrointestinal tract.
In humans, as well as in mice, food intake activates the vagus nerve and thus the gut-brain vagal axis. The team demonstrated that when mice enter the compulsive binge-eating phase, the level of peripheral endocannabinoids secreted by the stomach and intestine increases. This drives to the inhibition of the vagal axis, which in turn leads to a reduction in satiety, thus causing overeating. Similar to a vicious circle, binge-eating leads to a compulsive reward-driven feeding. On the contrary, the inhibition of the endocannabinoids receptor (called CB1 receptor) elicits the activation of satiety through the vagus nerve, thereby leading to both a reduction of reward processes and a decrease in compulsive eating behavior.
Towards a new treatment
Hence, foods rich in fat and sugar, once in the gastroinstetinal tract, would facilitate the secretion of endocannabinoids. Endocannabinoids may contribute to the eating disorders by interfering with the interoceptive gut-brain relay, therefore complexifying the physiological routes by which we decide to eat (taste and/or emotional aspects).
Previous clinical studies in humans sought to reduce appetite and body weight by using anti-satiety molecules which, by inhibiting endocannabinoid receptors, triggered the sensation of satiety, thus dampening food intake. However, these molecules had a major limitation: they passed the blood-brain barrier which protects the brain from what occurs externally. In doing so, they cause significant secondary side effects such as psychotic disorders, depression…
In the current study, the new treatment is designed to modulate the activity of the gut-brain vagal axis but do not cross the blood-brain barrier: they are called peripheral antagonists. This suggests a reduction in the side effects associated with the treatments already available. Moreover, this study reveals the importance of studying the functional connectivity between the body and the brain and the interoception processes as levers to act on certain metabolic but also psychiatric disorders. These results reveal interesting perspectives that still need to be studied in humans. If proved to be beneficial in humans, this would be a major breakthrough in health to curtail the development of food-associated pathologies such as obesity, diabetes…
Sources
Identification of an endocannabinoid gut-brain vagal mechanism controlling food reward and energy homeostasis – Chloé Berland, Julien Castel, Romano Terrasi, Enrica Montalban, Ewout Foppen, Claire Martin, Giulio G. Muccioli, Serge Luquet, Giuseppe Gangarossa
Contact press
presse@u-paris.fr
Read more
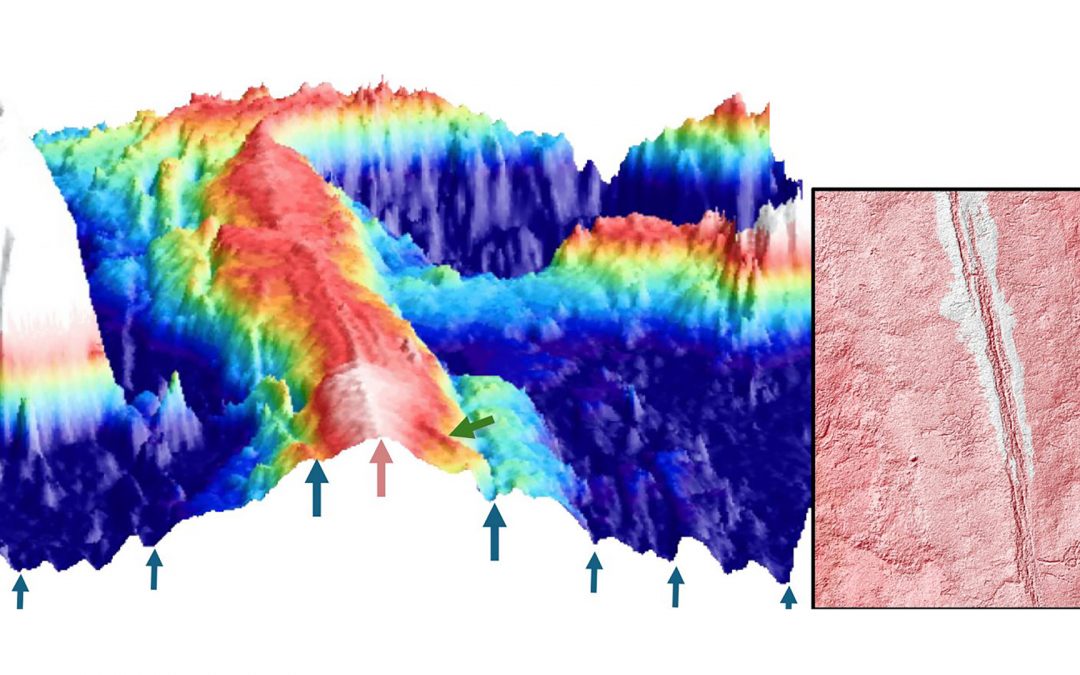
Evidence of magmatically induced faults at the East Pacific Rise
By comparison of ultra-high-resolution 3-D seismic imagery and bathymetry data collected at the East Pacific Rise (EPR) 9º50'N, researchers reveal the existence of magmatic induced faults near the ridge axis.

Convergences: A Strategic Alliance paving the Future of Biology-Health Research
A key milestone for the partnership between Institut Pasteur and Université Paris Cité, the first “Convergences” scientific seminar took place on June 17th at the university’s head office at Odéon.

Université Paris Cité receives 2 grants from Marie Sklodowska Curie Actions programme !
The results of the MSCA Staff Exchange 2023 call for proposals were published on May 28 2024. Université Paris Cité congratulates its two winners, Sylvain Chaty, Professor and Vice President for Culture, Science and Society at the AstroParticle and Cosmology...

MENTOR: Winner of the MSCA Doctoral Networks 2023
When abnormally activated, the mTOR protein can contribute to the development of cancers and certain monogenic diseases that affect around 2 million people worldwide. The multi-disciplinary MENTOR project, which stands for Metabolic control of cell growth by mTOR in...
