Scientists from the Institut Pasteur and the Vaccine Research Institute, in collaboration with KU Leuven (Leuven, Belgium), Orléans Regional Hospital, Hôpital Européen Georges Pompidou (AP-HP), Inserm and the CNRS, studied the sensitivity of the Omicron variant to monoclonal antibodies used in clinical practice to prevent severe forms of the disease in people at risk, as well as to antibodies in the blood of individuals previously infected with SARS-CoV-2 or vaccinated. They compared this sensitivity with that of the Delta variant. The scientists demonstrated that Omicron is much less sensitive to neutralizing antibodies than Delta. The scientists then analyzed the blood of people who had received two doses of the Pfizer or AstraZeneca vaccine. Five months after vaccination, the antibodies in the blood were no longer capable of neutralizing Omicron. This loss of efficacy was also observed in individuals who had been infected with SARS-CoV-2 within the past 12 months. Administering a booster dose of the Pfizer vaccine or a single vaccine dose in previously infected individuals led to a significant increase in antibody levels that was sufficient to neutralize Omicron. Omicron is therefore much less sensitive to the anti-SARS-CoV-2 antibodies currently used in clinical practice or obtained after two vaccine doses.
The study was published as a preprint on the bioRxiv website on December 15, 2021 and in Nature on Dec 23, 2021.
The Omicron variant was detected for the first time in South Africa in November 2021 and has since spread to many countries. It is expected to become the dominant variant within a few weeks or months.
Initial epidemiological studies demonstrate that the Omicron variant is more transmissible than the Delta variant. The Omicron variant’s biological characteristics are still relatively unknown. It has more than 32 mutations in the spike protein compared with the first SARS-CoV-2 and was designated as a variant of concern by WHO on November 26, 2021.
In South Africa, the Omicron variant replaced the other viruses within a few weeks and led to a sharp increase in the number of cases diagnosed. Analyses in various countries indicate that the doubling time for cases is approximately 2 to 4 days. Omicron has been detected in dozens of countries, including France and became dominant by the end of 2021.
In a new study supported by the European Union’s Health Emergency Preparedness and Response Authority (HERA), scientists from the Institut Pasteur and the Vaccine Research Institute, in collaboration with KU Leuven (Leuven, Belgium), Orléans Regional Hospital, Hôpital Européen Georges Pompidou (AP-HP) and Inserm, studied the sensitivity of Omicron to antibodies compared with the currently dominant Delta variant. The aim of the study was to characterize the efficacy of therapeutic antibodies, as well as antibodies developed by individuals previously infected with SARS-CoV-2 or vaccinated, in neutralizing this new variant.
The scientists from KU Leuven isolated the Omicron variant of SARS-CoV-2 from a nasal sample of a 32-year-old woman who developed moderate COVID-19 a few days after returning from Egypt. The isolated virus was immediately sent to scientists at the Institut Pasteur, where therapeutic monoclonal antibodies and serum samples from people who had been vaccinated or previously exposed to SARS-CoV-2 were used to study the sensitivity of the Omicron variant.
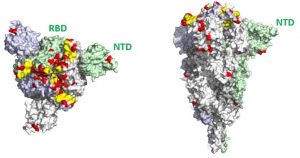
3D visualization of mutations in the spike protein of the Omicron variant. Left: overhead view. Right: lateral view. Mutations are indicated in red.They occur all over the spike protein but particularly in the receptor binding domain (RBD) and in the region known as the N-terminal domain (NTD).© Institut Pasteur – Félix Rey
The scientists used rapid neutralization assays, developed by the Institut Pasteur’s Virus and Immunity Unit, on the isolated sample of the Omicron virus. This collaborative multidisciplinary effort also involved the Institut Pasteur’s virologists and specialists in the analysis of viral evolution and protein structure, together with teams from Orléans Regional Hospital and Hôpital Européen Georges Pompidou in Paris.
The scientists began by testing nine monoclonal antibodies used in clinical practice or currently in preclinical development. Six antibodies lost all antiviral activity, and the other three were 3 to 80 times less effective against Omicron than against Delta. The antibodies Bamlanivimab/Etesevimab (a combination developed by Lilly), Casirivimab/Imdevimab (a combination developed by Roche and known as Ronapreve) and Regdanvimab (developed by Celtrion) no longer had any antiviral effect against Omicron. The Tixagevimab/Cilgavimab combination (developed by AstraZeneca under the name Evusheld) was 80 times less effective against Omicron than against Delta.
“We demonstrated that this highly transmissible variant has acquired significant resistance to antibodies. Most of the therapeutic monoclonal antibodies currently available against SARS-CoV-2 are inactive,” comments Olivier Schwartz, co-last author of the study and Head of the Virus and Immunity Unit at the Institut Pasteur.
The scientists observed that the blood of patients previously infected with COVID-19, collected up to 12 months after symptoms, and that of individuals who had received two doses of the Pfizer or AstraZeneca vaccine, taken five months after vaccination, barely neutralized the Omicron variant. But the sera of individuals who had received a booster dose of Pfizer, analyzed one month after vaccination, remained effective against Omicron. Five to 31 times more antibodies were nevertheless required to neutralize Omicron, compared with Delta, in cell culture assays. These results help shed light on the continued efficacy of vaccines in protecting against severe forms of disease.
“We now need to study the length of protection of the booster dose. The vaccines probably become less effective in offering protection against contracting the virus, but they should continue to protect against severe forms,” explains Olivier Schwartz.
“This study shows that the Omicron variant hampers the effectiveness of vaccines and monoclonal antibodies, but it also demonstrates the ability of European scientists to work together to identify challenges and potential solutions. While KU Leuven was able to describe the first case of Omicron infection in Europe using the Belgian genome surveillance system, our collaboration with the Institut Pasteur in Paris enabled us to carry out this study in record time. There is still a great deal of work to do, but thanks to the support of the European Union’s Health Emergency Preparedness and Response Authority (HERA), we have clearly now reached a point where scientists from the best centers can work in synergy and move towards a better understanding and more effective management of the pandemic,” comments Emmanuel André, co-last author of the study, a Professor of Medicine at KU Leuven (Katholieke Universiteit Leuven) and Head of the National Reference Laboratory and the genome surveillance network for COVID-19 in Belgium.
The scientists concluded that the many mutations in the spike protein of the Omicron variant enabled it to largely evade the immune response. Ongoing research is being conducted to determine why this variant is more transmissible from one individual to the next and to analyze the long-term effectiveness of a booster dose.
Source
Considerable escape of SARS-CoV-2 Omicron to antibody neutralization, Nature, 23 décembre 2021
Delphine Planas1,2*, Nell Saunders1,3*, Piet Maes4*, Florence Guivel-Benhassine1, Cyril Planchais5, Julian Buchrieser1, William-Henry Bolland1,3, Françoise Porrot1, Isabelle Staropoli1, Frederic Lemoine6, Hélène Péré7,8, David Veyer7,8, Julien Puech7, Julien Rodary7, Guy Baele4, Simon Dellicour4,9, Joren Raymenants10, Sarah Gorissen10, Caspar Geenen10, Bert Vanmechelen4, Tony Wawina -Bokalanga4, Joan Martí-Carreras4, Lize Cuypers11, Aymeric Sève12, Laurent Hocqueloux12, Thierry Prazuck12, Félix Rey13, Etienne Simon-Lorrière14, Timothée Bruel1,2,$, Hugo Mouquet5,$, Emmanuel André10,11,$ and Olivier Schwartz1,2,$
- Institut Pasteur, Université Paris Cité, CNRS UMR3569, Virus and Immunity Unit, 75015 Paris, France
- Vaccine Research Institute, 94000 Créteil, France
- Université Paris Cité, École doctorale BioSPC 562, 75013 Paris, France
- KU Leuven, Rega Institute, Department of Microbiology, Immunology and Transplantation, Laboratory of Clinical and Epidemiological Virology, Leuven, Belgium
- Institut Pasteur, Université Paris Cité, INSERM U1222, Humoral Immunology Laboratory, 75015 Paris, France
- Institut Pasteur, Université Paris Cité, CNRS USR 3756, Hub de Bioinformatique et Biostatistique, 75015 Paris, France.
- Hôpital Européen Georges Pompidou, Laboratoire de Virologie, Service de Microbiologie, 75015 Paris, France
- Centre de Recherche des Cordelier, INSERM, Université Paris Cité, Sorbonne Université, Functional Genomics of Solid Tumors (FunGeST), 75006 Paris, France
- Université Libre de Bruxelles, Spatial Epidemiology Lab (SpELL), Bruxelles, Belgium
- KU Leuven, Department of Microbiology, Immunology and Transplantation, Laboratory Clinical Microbiology, Leuven, Belgium
- University Hospitals Leuven, Department of Laboratory Medicine, National Reference Centre for Respiratory Pathogens, Leuven, Belgium
- CHR d’Orléans, service de maladies infectieuses, Orléans, France
- Institut Pasteur, Université Paris Cité, CNRS UMR3569, Structural Virology Unit, 75015 Paris, France
- Institut Pasteur, Université Paris Cité, CNRS UMR3569, G5 Evolutionary genomics of RNA viruses, 75015 Paris, France
*: co-first authors
$: co-last authors
DOI: 10.1038/d41586-021-03827-2
Read more

Call for projects 2025 UPCité – King’s College London
The call for projects between Université Paris Cité (UPCité) and our privileged partner King's College London (KCL), has been launched this friday, May 9th 2025. The objectives Université Paris Cité and King's College London are offering offering a seed funding for...

UM6P and UPCité Offer Two Joint PhD Scholarships
Mohammed VI Polytechnic University (UM6P) and Université Paris Cité (UPCité) are strengthening their collaboration by offering two joint PhD scholarships for thesis projects affiliated with one of UPCité’s Graduate Schools. This call aims to reinforce...

Radon Emissions on Mars and the Moon: The Contribution of Meteorites

Results of the first call for projects with Mohammed VI Polytechnic University
The first call for projects launched between Université Paris Cité and Université Mohammed VI Polytechnic in 2024 generated a great deal of interest. 24 proposals were submitted by pairs of researchers, one from UPCité and the other from UM6P, in all disciplines. A...
