In response to the thrombosis cases that occurred as a consequence of the Covid-19 vaccinations, Prof. David Smadja, professor of hematology at Université Paris Cité, member of the hematology department of the Georges-Pompidou European Hospital AP-HP and and his team at the Inserm Unit 1140 (Université Paris Cité), analyzed, at the request of the WHO Pharmacovigilance Center, more than 2,000 cases reported worldwide. The results of this study, published in the European Respiratory Journal, confirm the rarity of thrombosis cases.
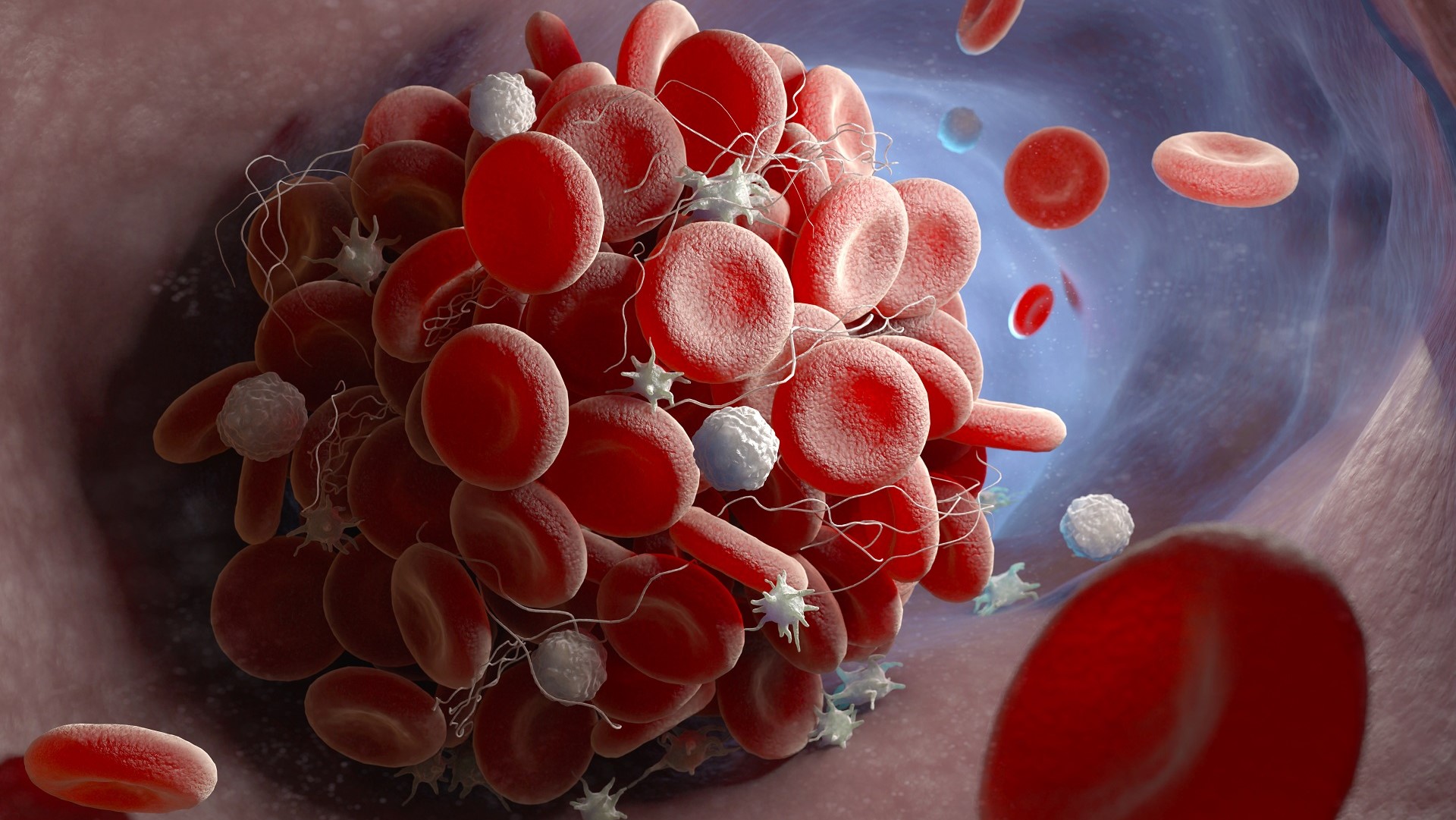
One of the most common complications of Covid-19 in its acute phase is a vascular disease that causes an activation of blood coagulation and thus the formation of blood clots at the origin of thrombosis, particularly in the lungs. Created more than a year ago, the Covid team (ANR-Fondation de France: SARCODO project), directed by Pr David Smadja, is specifically interested in coagulation and vascular disorders.
Prof. Smadja was approached by the World Health Organization (WHO) Pharmacovigilance Center, as soon as the first alerts of thrombosis cases in patients vaccinated with the AstraZeneca vaccine were reported. The team was asked to study and analyze thrombosis cases following vaccination with Pfizer, Moderna and AstraZeneca vaccines. The team analyzed and classified more than 2,000 cases of thrombosis reported worldwide by healthcare professionals to the WHO Pharmacovigilance Centre between December 15, 2020 and March 16, 2021.
Even though it is impossible to calculate an exact incidence (number of cases in relation to the number of vaccines administered) and insofar these are reports of adverse events, the initial results show that cases of venous and arterial thrombosis exist with the three vaccines. However, these thrombotic occurrences are in minute proportions, which does not allow us to conclude that there is an excess risk of thrombosis related to vaccines, regardless of the vaccine.
According to the study, it appears that the phenomenon of thrombosis could be linked in rare cases to an immune reaction by the body. This reaction may result in thrombocytopenia, a decrease in the number of platelets in the bloodstream. As of March 16th, 2021, the team had identified 1 case of thrombosis similar to what was already known about the phenomenon of allergic reaction to heparin (heparin thrombocytopenia) even though this patient had never received heparin. In this case, the decrease in the number of blood platelets (thrombocytopenia) is due to their aggregation together to form blood clots, resulting in thrombosis.
Typically prescribed as a blood thinner, heparin can provoke immune system reactions in some patients, leading to the formation of antibodies that cause the aggregation of blood platelets and promote the appearance of blood clots that cause thrombosis. This autoimmune process, which develops in some patients, is well known and managed medically by anticoagulants other than heparin and its derivatives.
Since March 16th, additional cases with these same side effects have been reported after having been vaccinated with adenovirus vaccines (AstraZeneca, Janssen). To date, more than fifty cases have been documented worldwide, which is still extremely rare given the number of people vaccinated.
It thus appears that there are very rare forms of thrombosis specifically related to adenovirus vaccines, although the causes and the specific mechanisms of action are not yet actually known. Medical management of these rare patients is now described and recommended by the International Society of Hemostasis and Thrombosis (ISTH).
Prof. Smadja points out that there is no specific risk factor, identified to date, which predisposes to post-vaccination side effects (see Recommendations of the COVID AP-HP working group). Further studies will be required to investigate the prevalence of hemostasis abnormalities associated with multiple thrombosis after adenovirus and RNA vaccines.
Despite the emergence of very rare side effects, Professor Smadja reiterates that the benefits of the vaccine in the fight against the COVID-19 epidemic far outweigh the risk of side effects.
Reference
Vaccination against COVID-19: insight fromarterial and venousthrombosis occurrence using data fromVigiBase. David M Smadja 1 2 , Qun-Ying Yue 3 , Richard Chocron 4 , Olivier Sanchez 2 5 , Agnes Lillo-Le Louet 6
- Université Paris Cité, Innovative Therapies in Hemostasis, INSERM, Paris, France; Hematology Department and Biosurgical Research Lab (Carpentier Foundation), Assistance Publique Hôpitaux de Paris-Centre (APHP-CUP), Paris, France.
- F-CRIN INNOVTE network, Saint Etienne, France.
- Uppsala Monitoring Centre, Uppsala, Sweden.
- Emergency Department, Université Paris Cité, PARCC, INSERM U970, AH-HP-Centre Université Paris Cité (APHP-CUP), Paris, France.
- Université Paris Cité, Innovative Therapies in Hemostasis, INSERM, Paris, France; Respiratory Medicine Department and Biosurgical Research Lab (Carpentier Foundation), Assistance Publique Hôpitaux de Paris-Centre (APHP-CUP), Paris, France.
- Université Paris Cité, Innovative Therapies in Hemostasis, INSERM, Paris, France; Pharmacovigilance Department, Assistance Publique Hôpitaux de Paris-Centre (APHP-CUP), Paris, France.
David Smadja is a professor of hematology at Université Paris Cité and a member of the hematology department of the Georges-Pompidou European Hospital AP-HP. He directs the Laboratoire de Recherches Biochirurgicales of the Carpentier Foundation and a research team in the Inserm 1140 unit at the Georges-Pompidou European Hospital AP-HP.
Contact : presse@u-paris.fr
Read more

The Indian Institute of Science Education and Research in Pune and the Indian Institute of Technology in Bombay: UPCité’s first partners in India.
Antoine Kouchner, Vice-President of Strategy and International Relations at Université Paris Cité, travelled to India for the official visit of the President, Emmanuel Macron, to the High-Level Scientific and Academic Meetings (RUSH). In this context, UPCité...

International Day of Women and Girls in Science: celebrating the women who push research forward
February 11 was the International Day of Women and Girls in Science. On this day, Université Paris Cité reaffirms its commitment to the equality between men and women and celebrates the journey of the women who advance research. Between celebrating our heritage and...

INC Day 2025: an international scientific day dedicated to neuroscience
The Neuroscience and Cognition Institute of Université Paris Cité (INC) organized a new edition of the INC Day, focused on neurodevelopmental trajectories. A key partner of the event, the Graduate School Neuroscience invited its first year and second year master...

Abraha and Pierre: A Friendship to Preserve the People’s Memory of War
In Paris, two historians’ paths crossed. One had just arrived from Ethiopia, carrying notebooks filled with daily observations written during the war in Tigray. The other, based in France, is a specialist of Ethiopian modern history. From this encounter, a partnership...
